Is Fog A Homogeneous Mixture
Heterogeneous and Homogeneous Mixture
"Let'south learn about what is mixture, homogeneous mixture, heterogeneous mixture with examples, solution, suspension, colloid, properties of mixture etc."
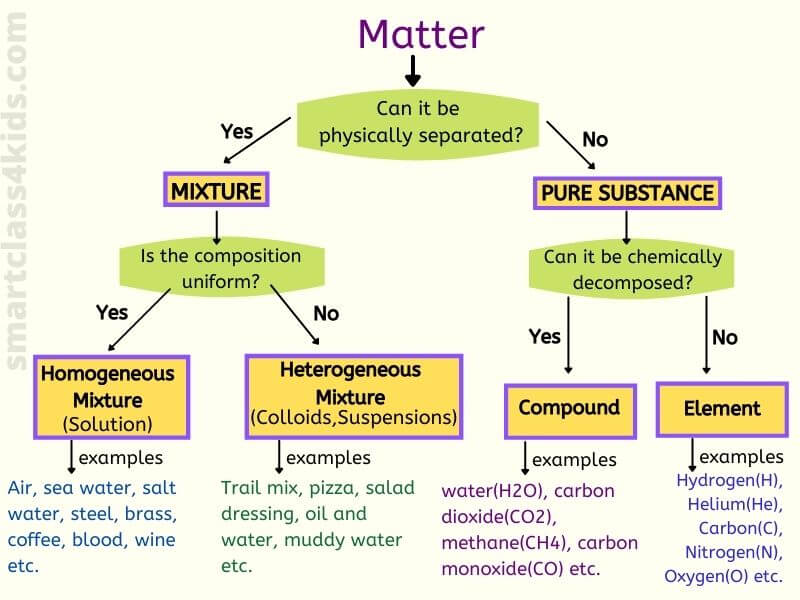
What is a mixture?
A mixture is a affair that consists of TWO or more types of substances mixed PHYSICALLY but Not chemically combined. Since mixtures are not chemically combined, we can carve up them by concrete methods. Mixtures can be solid, liquid, gas.
As well Read: Classifying Affair
Properties of mixture
In that location are 5 backdrop of mixtures listed beneath.
- The mixture has no stock-still proportion of the components.
- The substances of a mixture retain their properties.
- The mixture tin be separated past physical methods based on the physical properties of substances.
- No chemical process is involved in the formation of a mixture.
- The mixture has no stock-still melting points and boiling points.
Examples of a mixture
- Soda: a mixture of h2o, sugar, and flavorings.
- Fog – a mixture of water droplets and air
- Claret: A mixture of white blood cells, crimson blood cells, plasma and platelets
- Gasoline – a mixture of Petroleum, hydrocarbons, and fuel additives
- Crude oil: A mixture of organic compounds (mainly hydrocarbons)
- Seawater: A mixture of various salt and water.
- Air: a mixture of dissimilar gases like oxygen, carbon dioxide, nitrogen, argon, etc.
- Bleach: A mixture of chlorine, caustic soda, and water.
- Gunpowder: A mixture of sulfur, potassium nitrate, and carbon.
Types of mixture
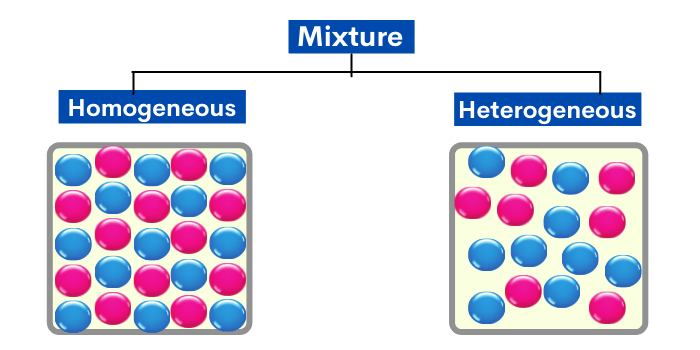
Mainly at that place are two types of mixtures: homogeneous mixture and heterogeneous mixtures.
What is a homogeneous mixture?
"Human" is a Greek word that means "aforementioned," "genus" ways "kind."
In this type of mixture, substances are so evenly mixed that information technology is difficult to distinguish one substance from another. It appears to incorporate only one kind of substance.
In uncomplicated words, If y'all can not see the different substances in a mixture, it's chosen homogeneous.
Types of homogeneous mixture?
- Solutions: A mixture formed when a substance (the solute) is dissolved in another substance (the solvent).
- Solute: the role of a solution that is being dissolved (unremarkably the bottom office).
Solvent: the function of a solution that dissolves the solute (usually the greater part).SOLUTION = SOLVENT + SOLUTE
Examples of homogeneous mixture
- A glass of lemonade (mixture of water, lemon juice, sugar, salt) is a homogeneous mixture because the dissolved sugar, salt, and lemon juice are evenly distributed throughout the entire sample. Yous tin't easily separate the lemon juice from the water; it's uniformly mixed.
- Stainless steel is fabricated from a homogeneous mixture of iron, chromium, and nickel.
- Air is a homogeneous mixture of nitrogen gas (78%), oxygen gas (21%), and modest amounts of various other gases.
- Oil, vinegar, dishwashing liquid, brass, wine, blood, seawater, natural gas, etc.
What is a heterogeneous mixture?
"Hetero" is a Greek word that means "different," "genus" means "kind."
In a heterogeneous mixture, substances are Non evenly mixed. The parts of these mixtures are noticeably different from one another.
In elementary words, If you can see the dissimilar substances in a mixture, it's called heterogeneous.
Types of heterogeneous mixture?
- Suspensions: a heterogeneous mixture in which the solute particles are so large that they settle out when left undisturbed for sometime. The particles of a suspension are visible to the naked eye. When pause are stirred, the particles seems to be evenly distibuted throughout the mixture. due east.g. sand-h2o, oil-h2o, grit particles in air etc.
- Colloids: a mixture in which the dispersed particles are medium in size between those of a solution and a pause. It means colloid is the intermediate state between solution and suspension. E.g. paints, milk, butter, fog etc.
Examples of a heterogeneous mixture
- sand and sugar
- salt and gravel
- ice cubes in a drink
- sand and water
- salt and oil
- oil and water
- orange juice with lurid
- craven noodle soup
- cereal and milk
- oatmeal and raisins
- salad
Characteristics of mixture
The major characteristics of mixtures are discussed below.
- Mixtures may be solid, liquid, or gas.
- Mixtures are impure substances.
- The constituents of a mixture practise not exist in a fixed ratio.
- A mixture is a combination of two or more than substances that are Non chemically combined.
- The substances may be separated by physical methods such as filtration, freezing, and distillation.
- They can either be heterogeneous or homogeneous in nature.
- There is no change in free energy during the formation of a mixture (energy is neither produced nor evolved).
- The mixture retains the properties of its components.
Divergence between homogeneous and heterogeneous mixture
Homogeneous
- mixed evenly
- These are chosen solutions
- Elective particles are not visible to the naked middle
- Separated chemically
- They represent the same concrete properties
- Homogeneous mixture examples are milk, oil, fog, seawater, etc.
Heterogeneous
- mixed unevenly
- These are called suspension/colloids
- Constituent particles are easily visible to the naked eye
- Separated physically
- They do not possess same concrete properties
- Heterogeneous mixture examples are sand water, salad, pizza, water-oil, salt-pepper, etc.
Is water a mixture?
No, water is not a mixture. H2o is a pure substance because it only contains one blazon of molecule, i.east., Water. Another point to think most is that a mixture is produced when ii substances are physically combined, non chemically. What would happen if you release hydrogen and oxygen from its cylinder in the surroundings? Practice you become water? No! You don't become water because you just mix hydrogen and oxygen molecules physically.
Just if y'all add together plenty free energy to the mixture of hydrogen and oxygen, then a chemical reaction occurs, and this way, water can be formed. So you can say that water is a pure substance and not a mixture.
Is air a mixture?
Yep, the air is a mixture because air can be separated into its constituents like nitrogen, oxygen etc., by the concrete process of partial distillation. Air has a variable composition, and it does not have a fixed formula well.
Is air a homogeneous mixture?
Yes, the air is a homogeneous mixture of nitrogen gas (78%), oxygen gas (21%), and pocket-sized amounts of diverse other gases. Information technology means, In the air, all gases would have a uniform composition. Therefore, the air is a homogeneous mixture.
Is soil a mixture?
Aye, the soil is a mixture of sand, clay, minerals, gases, liquids, and organic substances like dead constitute material and water.
Is rocky road ice foam a mixture?
Yes, a rocky road ice foam is an case of a heterogeneous mixture.
Is vinegar a mixture?
Yes, vinegar is a homogenous mixture of acetic acrid and water. As the mixture created has simply one stage, information technology is a solution. Through combining two or more chemical substances, mixtures are produced. If the result has more than one phase, it is a mixture; else, it is called a solution.
Is cough syrup a mixture?
Yes, cough syrup is an instance of a homogeneous mixture.
Is Fog A Homogeneous Mixture,
Source: https://smartclass4kids.com/homogeneous-mixture/
Posted by: burrowslifeatchas.blogspot.com


0 Response to "Is Fog A Homogeneous Mixture"
Post a Comment